Afr. J. Parasitol. Mycol. Entomol. , 2(1), 11; doi:10.35995/ajpme02010011
Hematological Changes among Malaria Patients Following Treatment with Artemisinin Derivatives in Nanoro, Burkina Faso
1
Institut de Recherche en Sciences de la Santé (IRSS)/Unité de Recherche Clinique de Nanoro (URCN), 11 BP 218 Ouagadougou CMS 11, Nanoro, Burkina Faso
2
Ministère de la Santé et de l’Hygiène Publique, Direction Régionale de la Santé et de l’Hygiène Publique du Centre-Ouest, Koudougou, Burkina Faso
3
Department of Biomedical and Public Health/Institut de Recherche en Sciences de la Santé (IRSS)/Unité de Recherche Clinique de Nanoro (URCN)
*
Corresponding author: berengerkabore@yahoo.fr; Tel.: +(00226)71947481
†
These authors contribute equally to this work (shared first co-authorship).
How to Cite: Kaboré, B.; Tahita, M.C.; Diallo, S.; Lompo, P.; Bognini, J.D.; Sawadogo, Y.E.; Sibidou, Y.; Kazienga, A.; Tinto, H. Hematological Changes among Malaria Patients Following Treatment with Artemisinin Derivatives in Nanoro, Burkina Faso. Afr. J. Parasitol. Mycol. Entomol. 2024, 2(1): 11; doi:10.35995/ajpme02010011.
Received: 11 May 2024 / Accepted: 30 July 2024 / Published: 20 August 2024
Abstract
:Background: Artemisinin and its derivatives are the key treatment for malaria, although cases of hematological toxicity have been reported. This study assessed the hematological toxicity of artemisinin derivatives using new parameters derived from a hematology analyzer. Methods: This is a secondary analysis from a diagnostic accuracy study of a new hematology analyzer for malaria diagnosis. Previous treatment and biological tests, including full blood count and malaria microscopy, were recorded. Participants were divided into three groups: (i) malaria-negative with no artemisinin treatment (Mal-/ACT-), (ii) malaria-negative with artemisinin treatment (Mal-/ACT+) and (iii) malaria-positive with artemisinin treatment (Mal+/ACT+). Erythropoiesis was assessed using reticulocyte absolute count (Ret-Ab) and the reticulocyte production index (RPI), and free hemoglobin level for hemolysis. Results: In total, data from 292 (31.0%) participants enrolled in the main study were used; 140 (47.9%) received artemisinin derivatives treatment at admission and 93 (31.8%) were malaria-positive. After multivariable analysis and adjustment for hemoglobin, age and C-reactive protein (CRP), the delta Ret-Ab (measuring the baseline and follow-up levels) did not significantly differ in the Mal+/ACT+ (β = 1.1, se(β) = 5.4, p = 0.8) and Mal-/ACT+ groups (β = 5.1, se(β) = 5.5, P = 0.4) compared to the Mal-/ACT- group. The same holds for the delta RPI. Free-Hb levels were elevated in the groups treated with ACT (Mal-/ACT+ and Mal+/ACT+) compared to Mal-/ACT-. However, this difference remained significant after adjustment for parasite density only for the Mal+/ACT+ group with a median delta free hemoglobin of 0.4 g/dL. Conclusion: There was no evidence of reticulocytopenia. However, post-treatment hemolysis with a limited impact was observed. Artemisinin and its derivatives remain a well-tolerated antimalarial drug.
Keywords:
malaria; artemisinin and its derivatives; erythropoiesis; hemolysis; anemiaBackground
Malaria remains a public health threat. Indeed, after an enthusiastic decrease followed by a small increase of 3% in 2020, the incidence rates have remained stable over the past 3 years. Globally, in 2022, there were an estimated 249 million cases, an increase of 5 million cases compared with 2021 [1]. Artemisinin and its derivatives, alone or in combination (artemisinin combination treatment: ACT), are currently the first-line treatment for severe and uncomplicated malaria infections [2]. The combinations comprise an artemisinin derivative that rapidly reduces the parasite biomass and gametocyte carriage [3,4], together with a slower-acting partner drug that clears the remaining parasites and provides post-treatment prophylaxis of which the duration depends on its pharmacokinetic properties [5,6,7]. Although artemisinins are generally safe, they can cause hematological toxicity. This is especially true for two types of adverse reactions: depression of erythropoiesis and delayed hemolysis [8,9].
In animal model and human studies, artemisinin and its derivatives (AD) were found to cause reticulocytopenia, a sign of decreased erythropoiesis [8,10,11,12]. Because reticulocyte response is affected by the level of anemia, the reticulocyte production index (RPI) may be a better erythropoiesis evaluation parameter. Furthermore, artemisinin derivatives have been linked to delayed hemolysis, which can be caused by the destruction of “pitted” erythrocytes or autoimmune reactions [9,13]. According to previous studies, this adverse reaction typically occurs 2 weeks after the onset of treatment in immune-naive patients with hyperparasitemia and may be severe, requiring a blood transfusion. However, these adverse reactions have been reported mainly from non-endemic areas (European countries and the USA), where some cases were considered severe. In malaria-endemic settings, however, AD’s hematologic toxicity is uncertain. Nonetheless, because anemia is also prevalent in these settings, this adverse reaction, if frequent, could contribute to worsening the already-precarious anemic status of malaria patients. Field studies from malaria-endemic areas [14,15], as well as studies in travelers [16,17], generally document hemolysis as low haptoglobin and high lactate dehydrogenase (LDH) levels combined with decreasing hemoglobin (Hb) levels. However, these parameters are not used in routine health practice in resource-limited settings such as malaria-endemic countries.
With the recent technology development, new hematology analyzers are equipped with a new detection capacity and reagents. These analyzers offer new parameters, allowing for a better characterization of blood cells and content, and thus, an overview of red blood cell turnover. Among these parameters, absolute reticulocyte counts (Ret-Ab) and the reticulocyte production index (RPI) allow for the assessment of erythropoiesis activity, while the free hemoglobin level helps measure hemolysis. Free hemoglobin (Free-Hb) levels were defined as the difference between standard Hb measurements (all red blood cells (RBC) are lysed) and optical Hb level (HGB-O), which measures Hb within intact RBCs.
In this study, we assessed the hematological toxicity of AD in a high-malaria-endemic region in Burkina Faso using a hematology analyzer named XN-1000 that measures the reticulocyte count (Ret-Ab), RPI and free hemoglobin levels. In contrast to previous studies, our study included a control group of subjects who were treated with artemisinin derivatives while no malaria was detected.
Materials and Method
Study Settings and Design
This is a secondary analysis of data from a prospective hospital-based diagnostic accuracy study (clinicalTrials.gov, NCT02669823), conducted at the Nanoro district hospital known as the ‘Centre Medical avec Antenne chirurgical’ (CMA) Saint Camille de Nanoro in Burkina Faso. The primary objective of the main study was to assess the diagnostic sensitivity and specificity of the XN-31 (new hematology analyzer) to detect malaria parasitemia in children and adults with an acute febrile illness compared to the thick blood smear microscopy as the gold standard or qPCR in the case of a negative thick smear or incongruent results of the XN-31 machine and microscopy [18] .
The Nanoro health district encompasses 1302 km2, with an estimated population of 188.837 in 2021 (Plan d’ Action 2021). Malaria is hyperendemic, with a peak during the rainy season (July–October) [19]. Almost all malaria cases are caused by Plasmodium falciparum, though Plasmodium ovale and Plasmodium malariae are also sporadically found [20]. As per the national guidelines for malaria management, all severe cases are treated with injectable artesunate or injectable quinine in pregnant women in the first trimester and/or ACT after the first trimester. Uncomplicated cases are treated with oral ACT or tableted quinine in pregnant women in the first trimester and/or ACT after the first trimester [21].
Study participants were enrolled between 23 March 2016 and 30 June 2017. Participants were recruited at the pediatric and general medicine outpatient departments of the “Centre Medical avec Antenne Chirurgicale” (CMA) Saint Camille Nanoro. Pregnant women were excluded from the study. Basic demographic and clinical data were recorded on a standardized case report form by qualified study nurses. Patients were followed daily during the hospitalization, and a definitive diagnosis was recorded at discharge. The follow-up was carried out approximately 14 days and 30 days after inclusion to assess if patients fully recovered, relapsed, or died.
Participant Selection and Classification
The current study participants were selected from the main study. Therefore, the inclusion and exclusion criteria were those from that study, of which the details were already published elsewhere [18]. Our analysis considered participants who were either positive or negative for malaria, and treated or not with artemisinin derivatives (injection and/or oral treatment). Participants were selected based on the availability of the full blood count (FBC) measurement at the study inclusion (baseline value) and during the follow-up, the malaria test result, AD treatment initiated at admission, hemoglobin (Hb) level of at least 5 g/dL at admission and no blood transfusion during hospitalization. The cut-off for the Hb level of at least 5 g/dL was chosen as below a Hb level is considered severe anemia with a potential blood transfusion indication, and therefore caused potential changes in the patient hematological parameters.
Participants were classified as follows: (i) malaria-microscopy-negative with no artemisinin derivative treatment (Mal-/ACT-), (ii) malaria-microscopy-negative with artemisinin derivative treatment (Mal-/ACT+) and (iii) malaria-microscopy-positive with artemisinin derivative treatment (Mal+/ACT+). In our settings, malaria was mainly diagnosed using HRP2 based RDT. Because of the persistence of the HRP2 antigen, some patients are still treated with AD while they are negative based on microscopy. This explains the cases of malaria-microscopy-negative treated with artemisinin derivatives (Mal-/ACT-). Ret-Ab and RPI were used as erythropoiesis indicators, and free-Hb for hemolysis. Free-Hb levels above the upper reference limit (i.e., typically ≥0.25 g/L in serum or ≥0.13 g/L in plasma), indicating hemolysis, although the estimate of free Hb from the Sysmex analyzer and the values given by Lippi et al. [22] may not be directly comparable. The assessment of the early effects of artemisinin derivatives were studied between AD administration and the 10-day follow-up (early onset), reflecting the effect on erythropoiesis. The data from subsequent periods were considered for the delayed effects (late onset), representing hemolysis.
Laboratory Procedures
Blood samples were collected in EDTA tubes and analyzed using the XN-1000 hematology analyzer from SYSMEX company, Kobe, Japan. The details of the study are described elsewhere [18].
Statistical Analysis
Data are reported as the median and interquartile range (IQR), unless stated otherwise. A multiple linear regression was conducted to assess the effect of artemisinin derivatives on the different outcome parameters. “Delta” parameters were created for Ret-abs, RPI and Free-Hb, indicating the difference between the follow-up and the baseline value of the respective parameters. Only one follow-up measurement was considered for each participant. In the case of more than one follow-up measurement, the latest was considered to minimize the disease effect on the parameters. A categorical variable for the different groups was defined and used in the regression analysis model, with the Mal-/ACT- as the reference group. Age, Hb, parasite density and inflammation (C-reactive protein: CRP) were adjusted. Analyses were performed using STATA 14 (Stata Corp., College Station, TX, USA), and a cut-off of 0.05 was used as the threshold for statistical significance. The correlation coefficients were calculated using the Spearman rank correlation method. The plots were made using R Statistical Software (v3.4.4) with the package ggplot2.
Ethics Statement
Written informed consent was obtained from all study participant, and the study protocol was approved by both the national ethics committee (ref. 2016-01-006) and the institutional review board of IRSS (ref. A03-2016/CEIRES) in Burkina Faso, the ethical committee of the University Hospital of Antwerp (ref. 15/47/492) and the internal review board of the Institute of Tropical Medicine Antwerp (ref. 1029/15).
Results
Study Population and General Characteristics
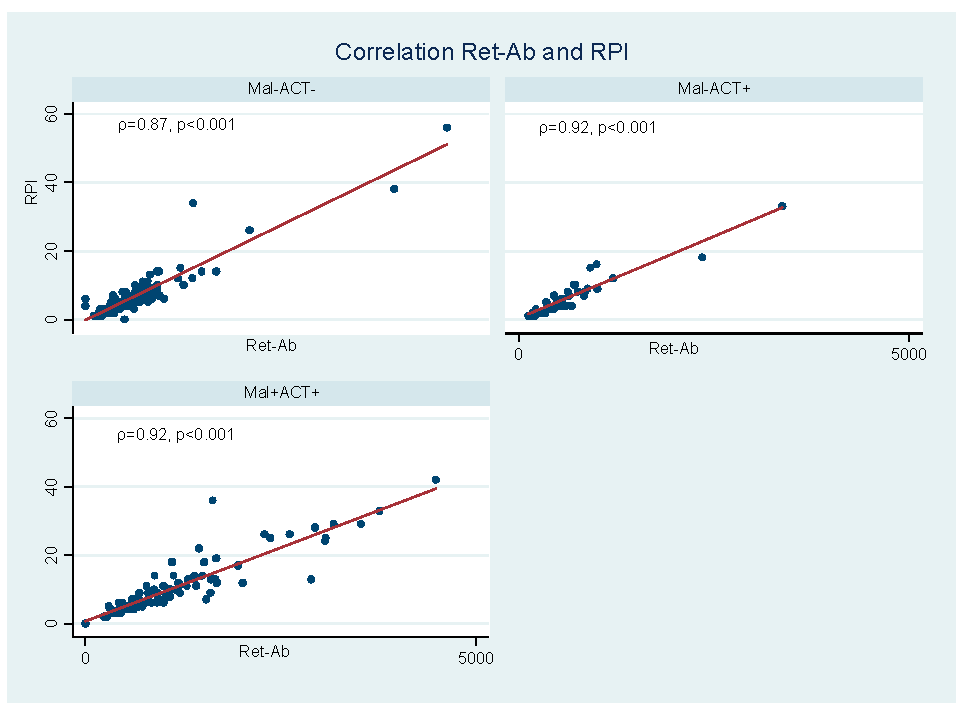
A total of 31.4% (292/930) participants participating in the main diagnostic accuracy study were enrolled in our study (Figure 1); 140/292 (47.9%) received artemisinin derivative treatment at admission and 93/292 (31.8%) had a positive malaria smear. Table 1 shows the demographic, clinical and biological characteristics of the study population at admission. The overall median age (range) was 4.8 (1.6–30.1) years old, with the Mal+/ACT+ group being the youngest, with a median age (range) of 2.9 (1.6–5.2) years, while the Mal-/ACT- group was significantly older (p = 0.001), with a median age (range) of 17.4 (1.7–43.6) years. The enrolment values of Ret-Ab (9.0 vs. 4.8 [Mal-/ACT-] and 4.9 [Mal-/ACT+]), RPI (0.8 vs. 0.5 [Mal-/ACT-] and 0.4 [Mal-/ACT+]) and plasma CRP (45.9 vs. 17.7 [Mal-/ACT-] and 19.2 [Mal-/ACT+]) concentrations were higher and hemoglobin (Hb) level (8.9 vs 10.4 [Mal-/ACT-] and 10.5 [Mal-/ACT+]) were lower for the Mal+/ACT+ group, whereas free-Hb did not significantly (p = 0.8) differ between the groups (0.1 in both groups treated with ACT). In all groups, Hb levels initially decreased and increased thereafter (Figure 2A). Ret-Ab and RPI values were highly correlated (ρ = 0.9, P = 0.0001) in the different patient groups (Figure 3).
Effect of ACT on Erythropoiesis
The values of Hb, Ret-Ab and RPI initially decreased in the Mal+/ACT+ group but increased, in contrast, in the Mal-/ACT+ group for Ret-Ab and RPI, while the Hb level also decreased (Figure 2A–C). In a multivariable analysis, after adjustment for Hb, age and CRP, the delta Ret-Ab in the early period was not significantly different in the Mal+/ACT+ (β = 1.1, se (β) = 5.4, P = 0.8) and Mal-/ACT+ groups (β = 5.1, se (β) = 5.5, P = 0.4) compared to the Mal-/ACT- group. In addition, the proportion of patients with a decreased delta Ret-Ab did not significantly differ between the groups, being 33% in the Mal-/ACT-, 22.6% in the Mal+/ACT+ and 19% in the Mal-/ACT+ group. The delta RPI levels were not significantly different during the early period in the Mal+/ACT+ (β = 0.3, se (β) = 0.6, P = 0.6), and Mal-/ACT+ groups (β = −0.2, se (β) = 0.6, P = 0.7) compared to the Mal-/ACT- group. Finally, the proportion of patients with a decreased delta RPI also did not differ between the groups.
Table 1.
Demographics, clinical and biological data at admission for all participants and by group.
| All Participants n = 292 | Mal-/ACT- n = 152 | Mal-/ ACT+ n = 47 | Mal+/ ACT+ n = 93 | P-Value | |
|---|---|---|---|---|---|
| Gender: F, n (%) | 173 (59%) | 87 (57.2%) | 29 (61.7%) | 57 (61.3) | -- |
| Age (years) | 4.8 (1.6–30.1) | 17.4 (1.7–43.6) | 3.3 (1.6–29.6) * | 2.9 (1.6–5.2) ** | =0.001 |
| Temperature, (°C) | 38.4 (38.0–39.4) | 38.3 (38–39) | 38.2 (37.8–39.5) | 38.8 (38–39.8) | NS |
| Weight (kg) | 14.7 (9.3–47.0) | 36.5 (9.7–51) | 16 (8.8–48)* | 10.7 (9.3–18) ** | *NS **=0.001 |
| Injectable artemisinin, n(%) | 184 (63%) | NA | 31 (66%) | 68 (73.1%) | NS |
| Parasite density, (number/ul) | NA | NA | NA | 26585 (1185–92269) | NA |
| CRP (mg/dl) | 29.6 (6.7–84.0) | 17.7 (4.0–72.1) | 19.2 (6.8–71.9) * | 45.9 (23.3–92.3) ** | *NS **=0.001 |
| RBC (106/ul) | 4.1 (3.4–4.6) | 4.1 (3.7–4.7) | 4.2 (3.8–4.8) * | 3.7 (2.8–4.3) ** | *NS **=0.001 |
| Hb (g/l) | 9.9 (8.2–11.2) | 10.4 (9.3–11.6) | 10.5 (8.2–11.6) * | 8.9 (7.0–10.6) ** | *NS **=0.001 |
| HCT (%) | 30.6 (25.5–34.3) | 31.8 (27.8–34.7) | 31.8 (26.6–35.9) * | 27.8 (21.1–32.0) ** | *NS **=0.001 |
| Ret-Ab, (104/ul) | 5.9 (3.7–8.8) | 4.8 (3.2–7.0) | 4.9 (3.1–7.2) * | 9.0 (6.0–14.3) ** | *NS **<0.001 |
| RPI (%) | 0.6 (0.4–0.9) | 0.5 (0.3–0.7) | 0.4 (0.2–0.8) * | 0.8 (0.5–1.3) ** | *NS **=0.001 |
| Free-Hb (g/dL) | 0.1 (−0.2–0.3) | 0.03 (−0.2–0.3) | 0.1 (−0.06–0.3) * | 0.1 (−0.2–0.4) ** | *NS **NS |
* P-value comparison between the Mal-/ACT+ and Mal-/ACT- groups; ** P-value comparison between the Mal+/ACT+ and Mal-/ACT- groups. Mal-/ACT-: malaria-microscopy-negative with no artemisinin derivative treatment; Mal-/ACT+: malaria-microscopy-negative with artemisinin derivative treatment; MAL+/ACT+: malaria-microscopy-positive with artemisinin derivative treatment; F: female; NA: not applicable; NS: non-significant; CRP: c-reactive protein; RBC: reb blood cells; Hb: hemoglobin; HCT: hematocrit; Ret-Ab: reticulocyte absolute count; RPI: reticulocyte production index; Free-Hb: free hemoglobin.
Figure 1.
Flow chart of participant selection. a: Only one case of malaria-positive received treatment other than artemisinin derivatives (Quinine). b: Cases with hemoglobin at admission less than 5 g/dL with a probable blood transfussion as recommended, but not properly captured in the database. AD = artemisinin derivatives; Mal+: malaria-microscopy-positive; Mal-: malaria-microscopy-negative; ACT+: artemisinin derivative treatment; ACT-: no artemisinin derivative treatment.
Figure 1.
Flow chart of participant selection. a: Only one case of malaria-positive received treatment other than artemisinin derivatives (Quinine). b: Cases with hemoglobin at admission less than 5 g/dL with a probable blood transfussion as recommended, but not properly captured in the database. AD = artemisinin derivatives; Mal+: malaria-microscopy-positive; Mal-: malaria-microscopy-negative; ACT+: artemisinin derivative treatment; ACT-: no artemisinin derivative treatment.

Figure 2.
A: Hemoglobin concentrations (Hb). B: Absolute reticulocyte numbers (Ret-Ab), C: reticulocyte production index (RPI), and D: free-Hb levels over time and in patients that are malaria-microscopy-negative with no ACT treatment (Mal-/ ACT-), malaria-microscopy-negative with ACT treatment (Mal-/ ACT+) and malaria-microscopy-positive with ACT treatment (Mal+/ ACT+). The vertical line on panels A to D indicates the cut-off point separating the early (until 10 days) and late (from 10 days and thereafter) period of the follow-up measurement.
Figure 2.
A: Hemoglobin concentrations (Hb). B: Absolute reticulocyte numbers (Ret-Ab), C: reticulocyte production index (RPI), and D: free-Hb levels over time and in patients that are malaria-microscopy-negative with no ACT treatment (Mal-/ ACT-), malaria-microscopy-negative with ACT treatment (Mal-/ ACT+) and malaria-microscopy-positive with ACT treatment (Mal+/ ACT+). The vertical line on panels A to D indicates the cut-off point separating the early (until 10 days) and late (from 10 days and thereafter) period of the follow-up measurement.

Figure 3.
Correlation between the reticulocyte absolute count (Ret-Ab) and reticulocyte production index (RPI) in the different groups of participants. This figure shows the correlation between the Ret-Ab and RPI in Mal-/ACT-, Mal-/ACT+ and Mal+/ACT+ groups. The RPI is positively correlated with the RPI, representing the bone marrow response to the decrease in the Hb level.
Figure 3.
Correlation between the reticulocyte absolute count (Ret-Ab) and reticulocyte production index (RPI) in the different groups of participants. This figure shows the correlation between the Ret-Ab and RPI in Mal-/ACT-, Mal-/ACT+ and Mal+/ACT+ groups. The RPI is positively correlated with the RPI, representing the bone marrow response to the decrease in the Hb level.
Effect of ACT on Free-Hb Levels
Free-Hb plasma levels above the reference range were found at baseline in 50.3 % of the Mal-/ACT- group, 55.6% of the Mal-/ACT+ group and 58.1% of the Mal+/ACT+ group. The values in the groups during the follow-up were 48.9%, 62.8% and 89.2%, respectively.
Free-Hb levels were elevated in the groups treated with ACT (Mal-/ACT+ and Mal+/ACT+) compared to the Mal-/ACT- group (Figure 1D). After adjusting for parasite density, the increase in free-Hb levels in the late period was significant in the Mal+/ACT+ group (β = 0.4, se (β)= 0.16, P = 0.02) compared to the Mal-/ACT- group, unlike in the Mal-/ACT+ group (β = 0.2, se (β) = 0.17, P = 0.2). Furthermore, 73 out of 87 (83.9 %) participants from the Mal+/ACT+ group had a higher free-Hb level in the late period compared to the baseline values with a median (IQR) delta free-Hb of 0.4 g/dL (0.2–0.6). Within the Mal+/ACT+ group, 26 of 73 patients (35.6%) also experienced concurrent lowering Hb levels: the median (IQR) Hb decrease (IQR) was 0.7 g/dL (0.2–1.4).
Discussion
Bone marrow depression and hemolysis may be caused by malaria or its treatment with artemisinin derivatives. The latter, in this study with a control population carried out in a high-transmission area, shows signs of hemolysis with no effects on the bone marrow.
The reticulocyte counts are an indicator of the erythropoietic activity of the bone marrow, while the RPI shows its capacity to compensate for anemia. Our results show that both parameters are increased in patients receiving artemisinin derivative treatment, which argues against artemisinin-derivative-mediated bone marrow depression. Our findings contrast with a review from Clark C. et al. [8], in which a Ret-Ab decrease was reported in 47–75% of healthy subjects and 0–34% in Asian malaria-positive patients. As our study was carried out in a high-malaria-transmission area (Burkina Faso), these differences may be due to genetic or acquired factors.
Our findings support the development of hemolysis as a feature of artemisinin and its derivatives, as first reported in Europeans patients by Zoller [16] and later confirmed in malaria-endemic areas [15,17]. The frequency of hemolysis was high (83.9%) in our study, especially compared to the 7% previously reported in severe-malaria patients in Gabon and Ghana [15] or 20–30% in non-immune patients in Germany [23]. Possibly, the difference in the measurement methods of hemolysis (using haptoglobin and high lactate dehydrogenase levels) may account for these differences. However, the severity of the hemolysis seemed limited as no notable decrease in Hb was noticed in our study. Our data on reticulocyte counts also indicate that the bone marrow capacity to compensate for anemia development is adequate, as reported before [23].
Some limitations of our study need to be highlighted. Firstly, we did not check for other possible causes of hemolysis, such as genetic red blood cells (sickle cell, thalassemia, G6PD) or autoimmune-related disorders. Secondly, the lack of several timepoints for the control did not allow us to assess the dynamic of the post-treatment hemolysis effect. Nonetheless, by using groups with different malaria statuses and artemisinin treatments, and a different time for the control timepoint, we were able to assess the effect of artemisinin with the control group of patients with no malaria and no artemisinin treatment.
Conclusion
In conclusion, the absence of reticulocytopenia in our study is not in favor of the negative effects of artemisinin and its derivatives on erythropoiesis. However, we found evidence of post-treatment hemolysis, but with a limited effect on hemoglobin levels. Therefore, artemisinin and its derivatives remain well-tolerated potent antimalaria drugs. Nonetheless, further studies with larger and more diverse populations should be carried out to check this particular aspect of the safety profile of artemisinin and its derivatives.
Author Contributions
B.K., M.C.T., S.D., P.L., J.D.B., Y.E.S., Y.S., A.K. and H.T. were involved in study design. B.K., S.D., J.D.B., Y.E.S., M.C.T. and Y.S. were responsible for study management in Burkina Faso. Analyses and writing were carried out by B.K., M.C.T. and A.K. All authors have read and approved the report before submission.
Funding
This work was supported by an unrestricted grant from SYSMEX Europe GmbH that supported the full funding of the presented studies. Furthermore, SYSMEX Europe GmbH provided the analyzers and technical assistance for running the analyzers. The study design, data collection, analysis and interpretation, as well as preparation of this report were completed independently.
Acknowledgment
The authors would like to thank the nurses from the “CMA Saint Camille de Nanoro”, the laboratory technicians from the Clinical Research Unit of Nanoro (CRUN), the team of data managers from CRUN and the study nurses from CRUN, Souleymane Ouedraogo and Alassane Compaore, for their dedication to the study. Furthermore, we thank all study participants for their participation.
Conflicts of Interest
All other authors report no potential conflicts of interest.
References
- World Malaria Report 2023; World Health Organization: Geneva, Switzerland, 2023.
- World Health Organization. Guidelines for the treatment of malaria, 2nd ed.; Geneva, Switzerland, 2010; p. 210. [Google Scholar]
- Hien, T.T.; White, N.J. Qinghaosu. Lancet 1993, 341, 603–608. [Google Scholar] [CrossRef] [PubMed]
- White, N.J. Qinghaosu (artemisinin): The price of success. Science 2008, 320, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Ashley, E.A.; White, N.J. Artemisinin-based combinations. Curr. Opin. Infect. Dis. 2005, 18, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Nosten, F.; White, N.J. Artemisinin-based combination treatment of falciparum malaria. Am. J. Trop. Med. Hyg. 2007, 77 (Suppl. 6), 181–192. [Google Scholar] [CrossRef] [PubMed]
- White, N.J. The role of anti-malarial drugs in eliminating malaria. Malar. J. 2008, 7 (Suppl. 1), S8. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.L. Effects of Artemisinins on Reticulocyte Count and Relationship to Possible Embryotoxicity in Confirmed and Unconfirmed Malarial Patients. Birth Defects Res. (Part A) Clin. Mol. Teratol. 2012, 94, 61–75. [Google Scholar] [CrossRef] [PubMed]
- Rehmana, K.; Lotsch, F.; Kremsner, P.G.; Ramharter, M. Haemolysis associated with the treatment of malaria with artemisinin derivatives: A systematic review of current evidence. Int. J. Infect. Dis. 2014, 29, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Effert, T.; Kaina, B. Toxicity of the antimalarial artemisinin and its derivatives. Crit. Rev. Toxicol. 2010, 40, 405–421. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.L. Embryotoxicity of the artemisinin antimalarials and potential consequences for use in women in the first trimester. Reprod. Toxicol. 2009, 28, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Slezakova, S.; Ruda-Kucerova, J. Anticancer Activity of Artemisinin and its Derivatives. Anticancer. Res. 2017, 37, 5995–6003. [Google Scholar] [PubMed]
- Arguin, P.M. Case definition: Postartemisinin delayed hemolysis. Blood 2014, 124, 167. [Google Scholar] [CrossRef] [PubMed]
- Fanello, C.; Onyamboko, M.; Lee, S.J.; Woodrow, C.; Setaphan, S.; Chotivanich, K.; Buffet, P.; Jauréguiberry, S.; Rockett, K.; Stepniewska, K.; et al. Post-treatment haemolysis in African children with hyperparasitaemic falciparum malaria; a randomized comparison of artesunate and quinine. BMC Infect. Dis. 2017, 17, 575. [Google Scholar] [CrossRef] [PubMed]
- Rolling, T.; Agbenyega, T.; Issifou, S.; Adegnika, A.A.; Sylverken, J.; Spahlinger, D.; Ansong, D.; Löhr, S.J.Z.; Burchard, G.D.; May, J.; et al. Delayed Hemolysis After Treatment With Parenteral Artesunate in African Children with Severe Malaria—A Double-center Prospective Study. J. Infect. Dis. 2013, 209, 1921–1928. [Google Scholar] [CrossRef] [PubMed]
- Zoller, T.; Junghanss, T.; Kapaun, A.; Gjørup, I.; Richter, J.; Hugo-Persson, M.; Mørch, K.; Foroutan, B.; Suttorp, N.; Yürek, S.; et al. Intravenous Artesunate for Severe Malaria in Travelers, Europe. Emerg. Infect. Dis. 2011, 17, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Rolling, T.; Agbenyega, T.; Krishna, S.; Kremsner, P.G.; Cramer, J.P. Delayed haemolysis after artesunate treatment of severe malaria e Review of the literature and perspective. Travel Med. Infect. Dis. 2015, 13, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Post, A.; Kaboré, B.; Reuling, I.J.; Bognini, J.; Van Der Heijden, W.; Diallo, S.; Lompo, P.; Kam, B.; Herssens, N.; Lanke, K.; et al. The XN-30 hematology analyzer for rapid sensitive detection of malaria: A diagnostic accuracy study. BMC Med. 2019, 17, 103. [Google Scholar] [CrossRef] [PubMed]
- Maltha, J.; Guiraud, I.; Kaboré, B.; Lompo, P.; Ley, B.; Bottieau, E.; Van Geet, C.; Tinto, H.; Jacobs, J. Frequency of Severe Malaria and Invasive Bacterial Infections among Children Admitted to a Rural Hospital in Burkina Faso. PLoS ONE 2014, 9, e89103. [Google Scholar] [CrossRef] [PubMed]
- Guiraud, I.; Post, A.; Diallo, S.N.; Lompo, P.; Maltha, J.; Thriemer, K.; Tahita, C.M.; Ley, B.; Derra, K.; Bottieau, E.; et al. Population-based incidence, seasonality and serotype distribution of invasive salmonellosis among children in Nanoro, rural Burkina Faso. PLoS ONE 2017, 12, e0178577. [Google Scholar] [CrossRef] [PubMed]
- Directives nationales de prise en charge du Paludisme; Programme National de Lutte contre le Paludisme; Ministere de la santé du Burkina Faso: Ouagadougou, Burkina Faso, 2021; p. 51.
- Lippi, G.; Giavarina, D.; Gelati, M.; Salvagno, G.L. Reference range of hemolysis index in serum and lithium-heparin plasma measured with two analytical platforms in a population of unselected outpatients. Clin. Chim. Acta 2014, 429, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Kurth, F.; Lingscheid, T.; Steiner, F.; Stegemann, M.S.; Bélard, S.; Menner, N.; Pongratz, P.; Kim, J.; von Bernuth, H.; Mayer, B.; et al. Hemolysis after Oral Artemisinin Combination Therapy for Uncomplicated Plasmodium falciparum Malaria. Emerg. Infect. Dis. 2016, 22, 1381–1386. [Google Scholar] [CrossRef] [PubMed]
© 2024 Copyright by Authors. Licensed as an open access article using a CC BY 4.0 license.

