Afr. J. Parasitol. Mycol. Entomol. 2023, 1(1), 2; doi:10.35995/ajpme1010002
Article
Spectrum and Antifungal Susceptibility Profiles of Candida Species Causing Vulvovaginitis in Lomé, Togo—Candida vaginitis and species involved in Lomé (Togo)
1
Faculté des Sciences de la Santé, Université de Lomé, Boulevard Eyadema, Lomé 01BP 1515, Togo; moklirita@gmail.com (A.M.M.); mireilleprincedavid@gmail.com (M.P.D.)
2
Division des Laboratoires du Ministère de la Santé, de l’Hygiène Publique et de l’Accès Universel aux Soins, 374, Avenue Georges Pompidou, Lomé 01BP 1161, Togo
3
Institut National d’Hygiène du Togo, 1, Rue Nangbéto, Quartier Administratif, Lomé 01BP 1396, Togo; yaogameli@yahoo.fr
4
Ecole Supérieure des Techniques Biologiques et Alimentaires, Université de Lomé, Boulevard Eyadema, Lomé 01BP 1515, Togo; cadjetey@yahoo.fr, fidelcab10@gmail.com
5
Centre Hospitalier Universitaire Sylvanus Olympio, 198, Rue de l’Hôpital, Lomé 01BP 57, Togo; mireilleprincedavid@gmail.com
*
Corresponding author: monicadork@yahoo.fr
How to Cite: Dorkenoo, A.M.; Afanyibo, Y.-G.; Adjétey-Toglozombio, A.-K.; Mokli, A.M.; Ataba, E.; Prince David, M. Spectrum and Antifungal Susceptibility Profiles of Candida Species Causing Vulvovaginitis in Lomé, Togo—Candida vaginitis and species involved in Lomé (Togo). Afr. J. Parasitol. Mycol. Entomol., 2023, 1(1): 2; doi:10.35995/ajpme1010002.
Received: 28 August 2022 / Accepted: 12 January 2023 / Published: 7 April 2023
Abstract
:Background: Vulvovaginal infections are common in women. In Togo’s medical biology laboratories, routine diagnosis is often limited to filamentation testing, and the identification of fungal species is rarely performed. The aim of this study was to update the spectrum of Candida species isolated in vulvovaginitis and to determine their antifungal susceptibility profiles. Methods: Cross-sectional analytical study was carried out in women presenting leukorrhea received in two public laboratories in Lomé from June 2015 to October 2015. Socio-demographic and clinical data were collected through a structured questionnaire during an individual interview. Vulvovaginal samples were cultured in Sabouraud dextrose agar with chloramphenicol. Identification of fungi was determined by filamentation test and API 20C AUX® gallery. The antifungal susceptibility profile was performed on five antifungal agents by a semi-solid medium microdilution technique of the ATB Fungus 3® gallery. Results: Candida albicans/dubliniensis/africana complex was isolated in 51.6% of cases by API 20C AUX® gallery. The same species were isolated by filamentation test in 12.3% of cases. All Candida species identified were sensitive to Voriconazole, whereas four species including Candida albicans/dubliniensis/africana complex had variable sensitivities compared to the five antifungal agents tested. Conclusions: This study demonstrated a high distribution of non-albicans species of Candida in Togo. Empirical treatment of candidiasis could contribute to increasing antifungal resistance. Routine fungi identification, at least by using an auxanogram test, could be instituted in medical laboratories to improve the therapeutic management of these diseases.
Keywords:
vulvovaginitis; Candida; API 20C AUX gallery; antifungal sensitivity; Lomé1. Introduction
Vulvovaginal infections are common among women during their active sexual and reproductive lives, and they are a major problem for these women regarding their health and comfort. Although usually mild, the symptoms of these infections are sometimes disabling. Recurrences are common, sometimes opportunistic, but have multiple contributing factors [1,2]. Most often, the causal agents are commensal fungi of vulvovaginal region. It results from the exacerbation of colonization, favoured by an imbalance in the normal flora and/or a decrease in immunity. The most identified fungi genus is Candida [3,4,5].
Identification of the fungal species involved in vulvovaginitis and the determination of their antifungal susceptibility profile are the key to good management of Candida species [6]. Causal agent identification is essential for regular updating of the epidemiological profile of circulating fungal species in general and particularly Candida species. It is necessary to establish an adequate algorithm for the treatment of vulvovaginitis, especially in low-income countries [7].
In Togo, routine diagnosis of vulvovaginal infections is rarely based on laboratory analysis; probabilistic treatment is the management rule adopted by clinicians. Moreover, when the examination is prescribed, the diagnostic approach for fungal infections in most microbiology laboratories is essentially based on the steps of direct examination, culture on Sabouraud-Chloramphenicol medium, and filamentation test. Antifungal susceptibility tests are performed only in exceptional cases [8]. This diagnostic procedure has limitations that could have a major impact on the treatment. In fact, treatment with an inappropriate antifungal agent could lead to an increase in recurrent cases and antifungal resistance phenomenon.
This study was carried out to help improve the laboratory diagnosis of infectious vulvovaginitis with the aim of identifying Candida species causing vulvovaginitis and determining their susceptibility profile to five antifungals.
2. Materials and methods
2.1. Study Design and Site
This was a cross-sectional study conducted from June 2015 to October 2015 at the microbiology laboratories of Institut National d’Hygiène (INH) and Centre Hospitalier Universitaire (CHU) de Lomé, which are both major reference laboratories in Togo.
2.2. Study Population and Sampling
All women experiencing leukorrhea with or without other symptoms visiting the microbiology laboratories of INH and CHU for mycological investigation were approached for recruitment into the study. Women who had received oral or topical antifungal therapy within the past 15 days were excluded.
2.3. Data Collection
Socio-demographic and clinical data were collected from consenting women during personal and confidential interviews using a structured questionnaire.
2.4. Laboratory Processes
2.4.1. Sample Collection
Using three sterile swabs with the patient’s identification on the sheaths, a sample was taken from the vaginal cavity, the cervix, and then from both the vaginal cavity and the cervix. The use of a projector lamp made it possible to appreciate the aspect of the vulva, the cavity, and the cervix as well as the characteristics of the leucorrhoea (colour, aspect, smell, abundance). After collection, the swabs were put back into the swab bags to avoid drying out. They were then sent directly to the laboratory for analysis.
2.4.2. Direct Microscopy and Culture
A high vaginal swab (HVS) sample was examined by direct microscopy with physiological saline for the presence of budding or non-budding yeasts, hyphae, or pseudohyphae. Another HVS sample was cultured in Sabouraud dextrose agar with chloramphenicol medium (Biokar Diagnostics, France) and incubated at 37 °C for 24 to 48 h [9]. Candida isolates were identified at the species level using the filamentation test and the API 20C AUX® gallery (BioMérieux, France).
The filamentation test was carried out by suspending a small portion of the isolated yeast colonies in physiological saline to obtain a fungal suspension with a turbidity of 0.5 McF. One calibrated handle loop (10 µL) of this suspension was added to 1 mL sheep serum and incubated at 37 °C for three hours. A drop of well-mixed incubated suspension was observed under the microscope and the presence or absence of a germ tube was noted.
The auxanogram was then performed using the API 20C AUX® gallery from the same colonies suspended in API C medium (BioMérieux, France) at a turbidity of 2 McF. The results were read by comparing with the growth controls, and fungi were identified using the API 20C AUX® gallery data sheet.
2.4.3. In Vitro Antifungal Susceptibility Testing
The antifungal susceptibility profiles of the fungi species isolated were determined by the microdilution method in semi-solid media of the ATB Fungus 3® gallery (BioMérieux, France). Five antifungals were tested: Amphotericin B (AMB), 5-Fluorocytosine (5-FC), Fluconazole (FCA), Itraconazole (ITR), and Voriconazole (VOR). A fungal suspension with a turbidity equivalent to 2 McF was prepared in a 2 mL ampule of API NaCl 0.85% medium (BioMérieux, France), and 20 µL of this suspension was homogenized in 7 mL of ATF Medium (BioMérieux, France). After that, 135 µL of this new suspension was inoculated into the 16 pairs of wells and incubated at 35 °C in a humid atmosphere for 24 h.
The appearance of turbidity in the wells, observed with the naked eye by placing the gallery on a black background, made it possible to determine the sensitivity of the fungi to the five antifungal agents tested. The interpretation took into account the growth score of each well with the control wells as references [10,11].
2.5. Ethical Considerations and Confidentiality
This study received ethical approval from the Bioethics Committee for Health Research of the Ministry of Health of Togo under the registration number N°293/2015/MSPS/CAB/SG/CBRS. In addition, each participant provided signed, written, informed consent, and the same procedure was applied to the parent or guardian of each child included in this study. The patient’s personal data were also kept in accordance with the rules of confidentiality and anonymity.
2.6. Statistical Analysis of the Data
Data analysis and graph design were performed by R version 4.0.5 software. Quantitative values were expressed either as mean ± standard deviation or as a unit or percentage.
3. Results
3.1. Characteristics of Study Participants
A total of 1647 women visited the two laboratories during the study period. Yeast was isolated from 410 (24.9%) of them, of which 122 signed the consent to participate in the study. The mean age of the study population was 29.9 ± 9.7 years, ranging from 12 to 62 years (Table 1). The most represented age groups were 25–34 years (36.1 %) and 15–24 years (32.8 %) (Table 1). Leucorrhoea was observed in 100% of the cases, whose characteristics are summarized in Table 1.
Table 1.
Distribution of patients according to age and macroscopic characteristics of vaginal swabs.
Table 1.
Distribution of patients according to age and macroscopic characteristics of vaginal swabs.
| Age of study population | ||
|---|---|---|
| N | % | |
| <15 | 2 | 1.64 |
| [15–24] | 40 | 32.79 |
| [25–34] | 44 | 36.07 |
| [35–44] | 28 | 22.95 |
| >45 | 8 | 6.56 |
| Total | 122 | 100.00 |
| Characteristics of leucorrhoea | ||
| Aspect | ||
| Thick | 90 | 73.77 |
| Fluid | 20 | 16.40 |
| Glutinous | 12 | 9.83 |
| Abondance | ||
| Scant | 20 | 16.40 |
| Abundant | 81 | 66.39 |
| Very abundant | 21 | 17.21 |
| Colour | ||
| Whitish | 73 | 59.84 |
| Brownish | 6 | 4.92 |
| Yellowish | 35 | 28.68 |
| Blood streaked | 8 | 6.56 |
| pH | ||
| Acid | 107 | 87.70 |
| Neutral | 15 | 12.30 |
| Basic | 0 | 0.00 |
| Odour | ||
| Odourless | 105 | 86.06 |
| Fetid | 17 | 13.94 |
3.2. Fungi Species Identified Distribution and Antifungal Susceptibility Profile
Direct examination with physiological saline showed blastospores in 72% of cases and associated with pseudohyphae in 18% of cases. Macroscopic examination of cultures showed white colonies which were smooth and creamy (52.5%), smooth and shiny (25.4%), creamy and wrinkled (12.3%), and fringed-edged (9.8%). Microscopic examination of colonies showed the presence of blastospores associated with pseudohyphae in 74.6% (91/122) of cases, and only blastospores in 25.4% (31/122) of cases.
The filamentation test was positive in 12.3% of cases (15/122). The API 20C AUX® gallery identified 100% of species of the Candida genus. The highest prevalences were observed for Candida albicans/dubliniensis/africana complex (51.6%), Candida glabatra (24.6%), Candida tropicalis (11.5%), and Candida krusei (9.8%). The C. albicans/dubliniensis/africana complex was the most identified fungus for all age groups, with a higher prevalence in the 15–24 years and 25–34 years age groups. C glabatra, C. tropicalis, and C. krusei were mostly isolated in the 25–34 years age group (Table 2). All species identified in the gallery API 20C AUX® showed pseudohyphae-associated blastospores on microscopic examination of the colonies, except C. glabrata, which presented only blastospores.
Table 2 shows the number and percentage of each fungal species isolated in the different age groups.
Table 2.
Fungi identified according to the age group.
| Age (years) | C. albicans * | C. glabrata | C. tropicalis | C. krusei | C. guillermondi | C. kefyr | C. parapsilosis | Total |
|---|---|---|---|---|---|---|---|---|
| <15 | 1 (0.8) | 0 (0.0) | 1 (0.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (1.6) |
| 15–24 | 29 (25.4) | 6 (4.9) | 2 (1.6) | 2 (1.6) | 0 (0.0) | 0 (0.0) | 1 (0.8) | 40 (32.8) |
| 25–34 | 19 (17.2) | 13 (10.6) | 7 (5.7) | 5 (4.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 44 (36.1) |
| 35–44 | 11 (9.0) | 8 (6.6) | 3 (2.5) | 4 (3.3) | 1 (0.8) | 1 (0.8) | 0 (0.0) | 28 (23.0) |
| >45 | 3 (2.5) | 3 (2.5) | 1 (0.8) | 1 (0.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 8 (6.6) |
| Total | 63 (51.6) | 30 (24.6) | 14 (11.5) | 12 (9.8) | 1 (0.8) | 1 (0.8) | 1 (0.8) | 122 (100) |
* Candida albicans/dubliniensis/africana complex. The results are presented in numbers (percentage).
3.3. Susceptibility Profile of Fungi Isolated
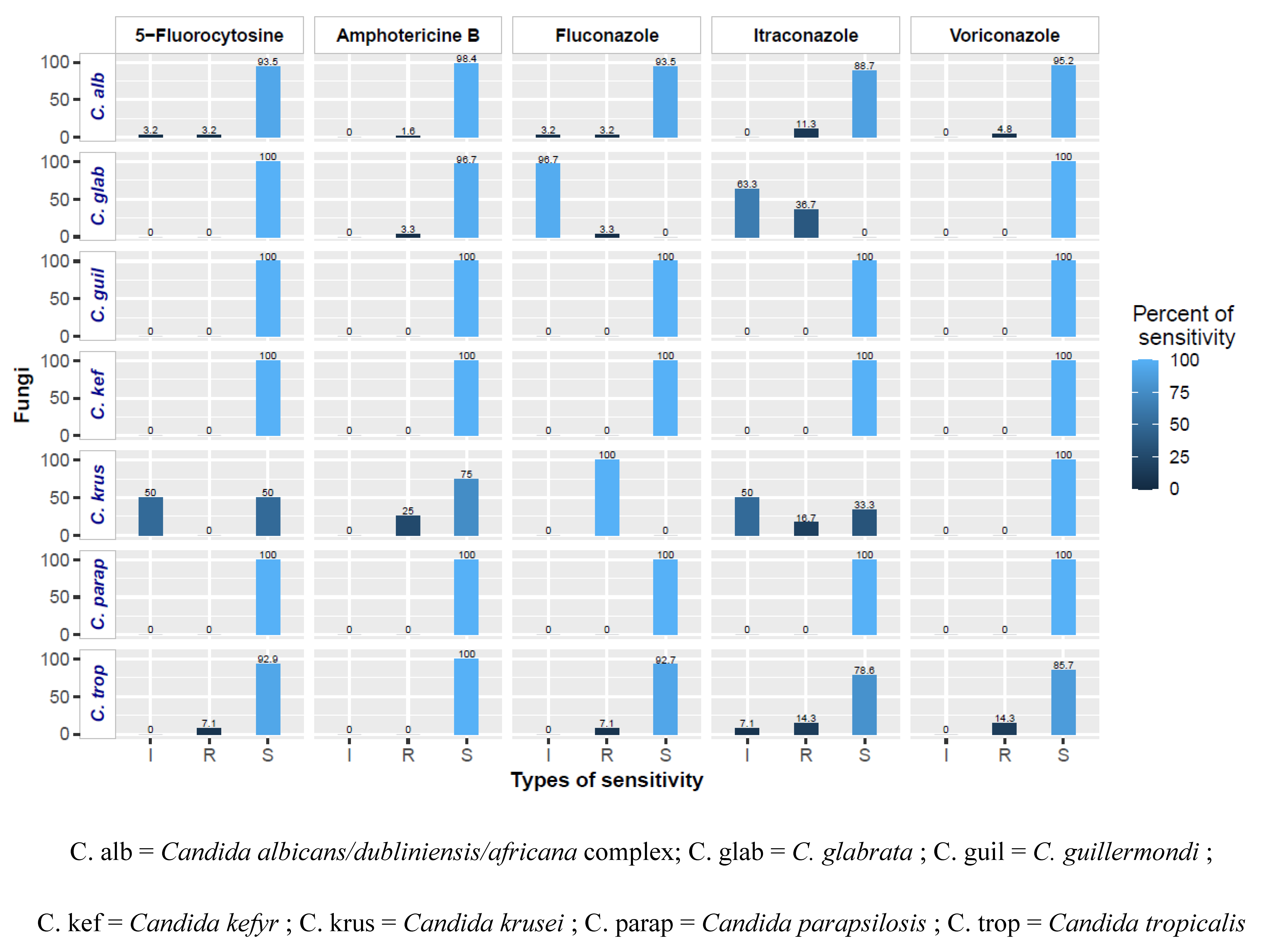
The sensitivity of species was different, depending on the antifungal agents tested. Candida guillermondi, Candida kefyr, and Candida prapsilosis showed high sensitivity to all antifungal agents tested. 5-Fluorocytosine had good antifungal activity on all isolated fungi except C. krusei. The different sensitivities are shown in Figure 1.
Figure 1.
Sensitivity of identified fungi to antifungal agents by MIC study.
4. Discussion
This study aimed to improve the laboratory diagnosis of infectious vulvovaginitis by identifying the Candida species responsible for vulvovaginitis and by determining their susceptibility profile to five antifungals. It sheds light on the management of vulvovaginitis whose diagnosis and treatment in Togo are mainly based on clinical presentations, in the absence of all laboratory diagnostic data.
The mean age of the study participants was 29.90 ± 9.70 years, corresponding to a sexually active adult population, similar to the mean age of 30.14 ± 11.14 of vulvovaginitis patients sampled by Agbo et al. [8] in Togo in 2015. The most represented age in our study is the range of 25–34 years (36.10%), also similar to that reported by Agbo et al. [8], but is in a lower proportion than that reported by Abia-Bassey and Utsalo [12] (65.20%) in Nigeria in 2006.
The presence of leucorrhoea and vaginal pruritus, found in the majority of participants in our study, was also reported by Develoux and Bretagne [13], Bohbot [14], Konate et al. [15], and Sylla et al. [16]. However, Waikhom et al. reported in 2020 that there was no association between these clinical manifestations and vulvovaginitis [17]. This simply explains that the clinical presentations of vulvovaginitis are not pathognomonic, as other vaginal conditions may cause these presentations [18,19]. It is therefore imperative to couple the clinical presentations with the results of the laboratory analysis in the diagnosis of vulvovaginitis [18], which is not the case to date in Togo.
C. albicans C. dubliniensis, and C. africana were the most isolated species, present in 51.6% of cases. This result is similar to that found by Dieng et al. [9] in Dakar, Senegal, in 2012, who found a prevalence of 52.75% by filamentation and auxanogram tests on samples with vaginal swabs accounting for 95.06% of cases. Konaté et al. found a higher prevalence of 82.50% in Abidjan, Côte d’Ivoire, by the same methods [15]. Candida species other than the C. albicans/dubliniensis/africana complex were also identified in our study: C. glabrata (24.60%), C. tropicalis (11.50%) and C. krusei (9.83%) for a total of 45.93%. The same findings were made in 2015 in Togo by Agbo et al. [8], who isolated 42.50% of non-albicans strains, and by Kengne et al. in Cameroon, who isolated 54.30% of strains from vulvovaginal infections [20]. The prevalence of C. albicans/dubliniensis/africana complex by filamentation test (12.30%) was much lower than that obtained by API 20C AUX® gallery (51.6%), demonstrating the limitation of this test in identifying all cases of C. albicans/dubliniensis/africana complex. Abia-Bassey and Utsalo in Nigeria [12] and Konaté et al. in Côte d’Ivoire [15] also noted a high prevalence of 70.70% and 82.50%, respectively, for this complex, using the API 20C AUX® gallery.
In neighbouring Ghana, Waikhom et al. found 74.1% of non-albicans Candida predominated by C. glabrata [17]. In their context, this could be the result of self-medication and prolonged antifungal treatment, which could lead to the selection of C. glabrata over C. albicans due to their increased resistance to commonly used and over-the-counter antifungal agents [18,21]. In addition, subpartite transmission of Candida spp. from the mother’s vagina to the newborn is reported. This can lead to widespread fungal infection in babies, especially in premature newborns, due to their immature immunity [22]. Therefore, pregnant women with vulvovaginitis in the first and second trimesters are likely to suffer from Candida chorioamnionitis, which can lead to abortion [42]. Vulvovaginitis can also lead to low birth weight and premature babies [23,24]. It is therefore imperative to establish a proper diagnosis of vulvovaginitis followed by adequate treatment, especially in women in general and pregnant women in particular [22].
The antifungal susceptibility profiles of the identified Candida species showed variable susceptibilities. Indeed, 5-Fluorocytosine (5-FC) showed good antifungal activity against all Candida isolates, except C. krusei, on which it induced an intermediate to 50% sensitivity. C. krusei showed a 25% resistance against Amphotericin B (AMB). Agbo et al., whose study was also conducted in Togo, also found a similar result using the solid-state diffusion method [8]. Apart from the natural resistance of C. krusei to Fluconazole (FCA), C. glabrata recorded an intermediate resistance rate of 96.2%, in contrast to the sensitivity of 65.60% reported by Bonouman-Ira et al. [7]. These differences in susceptibility may be explained by the fact that Bonouman-Ira et al. [7] used the disk diffusion method to determine susceptibility to antifungals. Itraconazole (ITR) and fluconazole were the only molecules inactive on C. glabrata compared to the other antifungals tested. The low susceptibility of fungi (C. glabratra and C. krusei) to FCA is thought to be related to the widespread use of this prophylactic antifungal agent in immunocompromised patients [25,26]. Voriconazole, on the other hand, was the molecule that showed high sensitivity for all isolated Candida species, confirming the results of Smith et al. [26], who noted a 100% sensitivity of all these fungi to Voriconazole. This result suggests that the drug of choice for effective management of vulvovaginal mycoses is Voriconazole, to which, unfortunately, access is limited in Africa, especially in Togo, in which, of the five antifungal drugs tested, only Fluconazole is commercially available and relatively affordable.
5. Conclusions
Our results showed that Candida albicans remains the most common species among the Candida genus isolated in vulvovaginitis in Lomé. The auxanogram is a more accurate method than the routine filamentation test for better identification of Candida spp. However, an important prevalence of non-albicans Candida species was noted. Voriconazole remains the most sensitive molecule for all isolated Candida species, although its pharmaceutical presentation is not yet available in Togo. Furthermore, molecular characterization of these species will give more details and other particularities of Candida spp. It is important to have these antifungals on the list of essential drugs in Togo for better management of fungal diseases.
Author Contributions
A.M.D. and A.M.M. designed the study; A.M.M. and A.K.A. conducted the laboratory work; M.P. oversaw the work; M.P. and A.M.D. supervised the laboratory work; A.M.M. and Y.A. cleaned and analysed the data; Y.A. and A.K.A. drafted the manuscript; A.M.D., E.A. and M.P. improved the draft. All authors read and approved the final version of the manuscript.
Funding
This research did not receive any specific grant from funding agencies.
Acknowledgments
We would like to thank the directors and staff of the two microbiology laboratories, and to all the women who agreed to participate in this study. Special thanks to Gani Kondi Tagbé (in memoriam) of the Institut National d’Hygiène, Katawa Gnatoulma from the University of Lomé and Sissinto-Savi de Tove Yolande of the University Hospital National Center of Cotonou in Benin for their invaluable contribution.
Conflicts of Interest
The authors declare no conflicts of interest. Approval for this study was received from the Togo MoH Bioethics Committee for Health Research. Written informed consent was obtained from the parent or guardian of each child enrolled in this study.
Availability of data and material
The datasets used and analysed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
AMB: Amphotericin B; CHU: University hospital; FCA: Fluconazole; 5-FC: 5-flurocytosine; HVS: high vaginal swab; INH: public health laboratory; ITR: Itraconazole; McF: McFarland; VOR: Voriconazole.
References
- Amouri, I.; Abbes, S.; Sellami, H.; Makni, F.; Sellami, A.; Ayadi, A. La candidose vulvovaginale. J. Mycol. Med. 2010, 20, 108–115. [Google Scholar] [CrossRef]
- Sobel, J. Pathogenesis of Candida vulvovaginitis. In Current Topics in Medical Mycology; McGinnis, M.R., Borgers, M., Eds.; Springer: New York, NY, USA, 1989; pp. 86–108. [Google Scholar] [CrossRef]
- Makni, F.; Sellami, A.; Trabelsi, H.; Sellami, H.; Cheikhrouhou, F.; Neji, S.; Ayadi, A. Évolution de la flore des levures isolées au CHU de Sfax, Tunisie. J. Mycol. Med. 2010, 20, 42–47. [Google Scholar] [CrossRef]
- Conley, L.J.; Ellerbrock, T.V.; Bush, T.J.; Chiasson, M.A.; Sawo, D.; Wright, T.C. HIV-1 infection and risk of vulvovaginal and perianal condylomata acuminata and intraepithelial neoplasia: A prospective cohort study. Lancet 2002, 359, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Durand, B. Traitements des Mycoses Vulvo-Vaginales Aiguës Et Récidivantes; Université de Lorraine: Thionville, France, 2017; Available online: https://hal.univ-lorraine.fr/hal-01932295 (accessed on July 11 2020).
- Choukri, F.; Benderdouche, M.; Sednaoui, P. In vitro susceptibility profile of 200 recent clinical isolates of Candida spp. to topical antifungal treatments of vulvovaginal candidiasis, the imidazoles and nystatin agents. J. Mycol. Med, 2014, 24, 303–307. [Google Scholar] [CrossRef]
- Bonouman-Ira, V.; Angora, E.; Djohan, V.; Vanga-Bosson, H.; Sylla-Thanon, K.; Beourou, S.; Koné, M. Profil de résistance des Candida non albicans à Abidjan en 2011. Revue Bio-Africa-N 2011, 9, 25–29. [Google Scholar]
- Agbo, Y.; Kpotsra, A.; Dorkendo, A.; Dogbe, H.; Tigossou, S.; Agbo, K. Que prescrire dans les candidoses vaginales au Togo? What do we prescribe in vaginal candidiasis in Togo? J. Rech. Sci. 2015, 17, 243–249. [Google Scholar]
- Dieng, Y.; Sow, D.; Ndiaye, M.; Guichet, E.; Faye, B.; Tine, R.; Lo, A.; Sylla, K.; Abiola, A.; Dieng, T. Identification of three Candida africana strains in Senegal. J. Mycol. Med. 2012, 22, 335–340. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Xiao, M.; Kudinha, T.; Mao, L.L.; Zhao, H.R.; Xu, Y.C. The widely used ATB FUNGUS 3 automated readings in China and its misleading high MICs of Candida spp. to azoles: Challenges for developing countries’ clinical microbiology labs. PLoS ONE 2014, 9, e114004. [Google Scholar] [CrossRef]
- Kouadio-Yapo, C.; Aka, N.; Bonouman-Ira, A.; Dou, G.; Sonan, K.; Zika, K.; Adoubryn, K.; Dosso, M. Resistance of Candida albicans to antifungal drugs in Abidjan (Cote dIvoire). Afr. J. Microbiol. Res. 2020, 14, 572–578. [Google Scholar] [CrossRef]
- Abia-Bassey, L.; Utsalo, S. Yeast associated with human infections in south-eastern Nigeria. Mycoses 2006, 49, 510–515. [Google Scholar] [CrossRef]
- Develoux, M.; Bretagne, S. Candidoses et levuroses diverses. J. EMC-Maladies Infectieuses 2005, 2, 119–139. [Google Scholar] [CrossRef]
- Bohbot, J.-M. Les sécrétions vaginales. J. Pelvi-Périnéologie 2008, 3, 19–24. [Google Scholar] [CrossRef]
- Konate, A.; Yavo, W.; Kassi, F.; Djohan, V.; Angora, E.; Barro-Kiki, P.; Bosson-Vanga, H.; Soro, F.; Menan, E. Aetiologies and contributing factors of vulvovaginal candidiasis in Abidjan (Cote d’Ivoire). J. Mycol. Med. 2014, 24, 93–99. [Google Scholar] [CrossRef]
- Sylla, K. Candidoses vulvo-vaginales au laboratoire de parasitologie-mycologie du Centre hospitalier universitaire de Fann, Dakar (Sénégal). RAMReS 2018, 5, 2. [Google Scholar]
- Waikhom, S.D.; Afeke, I.; Kwawu, G.S.; Mbroh, H.K.; Osei, G.Y.; Louis, B.; Deku, J.G.; Kasu, E.S.; Mensah, P.; Agede, C.Y.; et al. Prevalence of vulvovaginal candidiasis among pregnant women in the Ho municipality, Ghana: Species identification and antifungal susceptibility of Candida isolates. BMC Pregnancy Childbirth 2020, 20, 266. [Google Scholar] [CrossRef]
- Sobel, J.D.; Faro, S.; Force, R.W.; Foxman, B.; Ledger, W.J.; Nyirjesy, P.R.; Reed, B.D.; Summers, P.R. Vulvovaginal candidiasis: Epidemiologic, diagnostic, and therapeutic considerations. Am. J. Obstet. Gynecol. 1998, 178, 203–211. [Google Scholar] [CrossRef]
- Schaaf, V.M.; Perez-Stable, E.J.; Borchardt, K.J.; AoI, M. The limited value of symptoms and signs in the diagnosis of vaginal infections. Arch. Intern. Med. 1990, 150, 1929–1933. [Google Scholar] [CrossRef]
- Kengne, M.; Shu, S.V.; Nwobegahay, J.M.; Achonduh, O. Antifungals susceptibility pattern of Candida spp. isolated from female genital tract at the Yaoundé Bethesda Hospital in Cameroon. Pan Afr. Med. J. 2017, 28, 294. [Google Scholar] [CrossRef]
- Spinillo, A.; Nicola, S.; Colonna, L.; Marangoni, E.; Cavanna, C.; Michelone, G. Frequency and significance of drug resistance in vulvovaginal candidiasis. Gynecol. Obstet. Investig. 1994, 38, 130–133. [Google Scholar] [CrossRef]
- Blaschke-Hellmessen, R. Subpartale Übertragung von Candida und ihre Konsequenzen: Subpartal transmission of Candida and its consequences. J. Mycoses. 1998, 41, 31–36. [Google Scholar] [CrossRef]
- Leli, C.; Mencacci, A.; Meucci, M.; Bietolini, C.; Vitali, M.; Farinelli, S.; D’Alò, F.; Bombaci, J.; Perito, S.; Bistoni, F. Association of pregnancy and Candida vaginal colonization in women with or without symptoms of vulvovaginitis. Minerva Ginecol. 2013, 65, 303–309. [Google Scholar] [PubMed]
- Holzer, I.; Farr, A.; Kiss, H.; Hagmann, M.; Petricevic, L. The colonization with Candida species is more harmful in the second trimester of pregnancy. Arch. Gynecol. Obstet. 2017, 295, 891–895. [Google Scholar] [CrossRef] [PubMed]
- George, G.; Cawood, C.; Puren, A.; Khanyile, D.; Gerritsen, A.; Govender, K.; Beckett, S.; Glenshaw, M.; Diallo, K.; Ayalew, K. Evaluating DREAMS HIV prevention interventions targeting adolescent girls and young women in high HIV prevalence districts in South Africa: Protocol for a cross-sectional study. J. BMC Women’s Health 2020, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Safdar, N.; Knasinski, V.; Simmons, W.; Bhavnani, S.; Ambrose, P.; Andes, D. Voriconazole therapeutic drug monitoring. Antimicrob. Agents Chemother. 2006, 50, 1570–1572. [Google Scholar] [CrossRef] [PubMed]
© 2023 Copyright by Authors. Licensed as an open access article using a CC BY 4.0 license.

